
Trig is an award-winning innovation and design firm that offers clients guided experiences in building a culture of innovation, understanding their customer needs, designing successful products, and developing lasting brands.
Our comprehensive innovation process customizes to your company vision and culture. We provide education, coaching, and expert specialists to work seamlessly across phases of innovation programs. Trig has expertise in human factors, customer research, brand, and design. We build solutions that resonate with people.

Craft your design strategy with novel insights and thoughtful virtual ideation to arrive at meaningful solutions. A strong foundation with a well-researched proprietary understanding of the customer will help you to establish expertise.

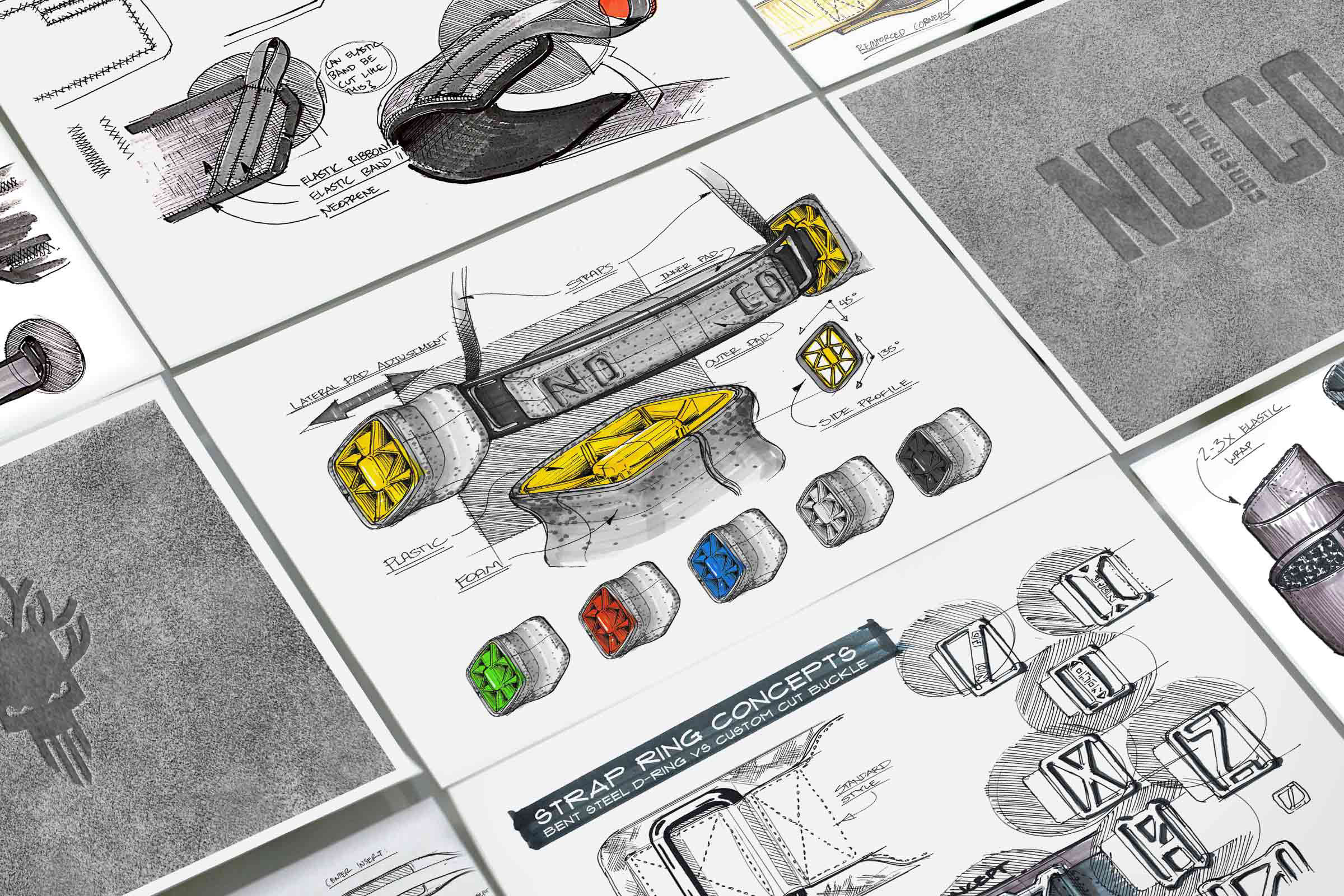
Successful new designs make an impact on your customer that goes past the first impression. We help our clients to create solutions that are human-centered, practical and eye-catching. We work closely with our clients through the many design choices that lead to exciting outcomes.


Crafting the perfect brand message requires discovering your brand’s story and telling it in a way that draws people in and leaves them craving for more. The clearer your voice and the more consistent your message, the more you will attract loyal customers who identify with your brand.

Trig specializes in all aspects of the design process; insights and ideation, design and development, and brand & digital experiences.
The inspiration for creating DHF Ready Ideation was to develop a toolset intended to support the creative work done prior to implementing design controls, while at the same time, capturing it, thus streamlining this transition. The results have not just met, but surpassed that original intention. DHF Ready Ideation provides a platform to increase business success, user satisfaction, and minimize potential harms by encouraging innovators and designers to consider a holistic view of medical device product development and encourage an evolutionary iterative process along the entire journey.


At Trig we aspire to serve as leaders in thought when it comes to all areas of design. Take a deeper dive into our featured posts to learn more about what makes us tick.